 Corrosion of reinforcing steel and other embedded metals is the leading cause of deterioration inconcrete. When steel corrodes, the resulting rust occupies a greater volume than the steel. This expansion creates tensile stresses in the concrete, which can eventually cause cracking, delamination, and spalling.
Corrosion of reinforcing steel and other embedded metals is the leading cause of deterioration inconcrete. When steel corrodes, the resulting rust occupies a greater volume than the steel. This expansion creates tensile stresses in the concrete, which can eventually cause cracking, delamination, and spalling.
Steel corrodes because it is not a naturally occurring material. Rather, iron ore is smelted and refined to produce steel. The production steps that transform iron ore into steel add energy to the metal.
Steel, like most metals except gold and platinum, is thermodynamically unstable under normal atmospheric conditions and will release energy and revert back to its natural state—iron oxide, or rust. This process is called corrosion.
For corrosion to occur, these elements must be present:
- There must be at least two metals (or two locations on a single metal) at different energy levels
- an electrolyte
- a metallic connection

In reinforced concrete, the rebar may have many separate areas at different energy levels. Concrete acts as the electrolyte, and the metallic connection is provided by wire ties, chair supports, or the rebar itself.
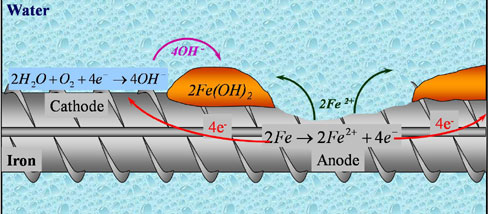
Corrosion is an electrochemical process involving the flow of charges (electrons and ions). At active sites on the bar, called anodes, iron atoms lose electrons and move into the surrounding concrete as ferrous ions. This process is called a half-cell oxidation reaction, or the anodic reaction, and is represented as:
2Fe → 2Fe2+ + 4e-
The electrons remain in the bar and flow to sites called cathodes, where they combine with water and oxygen in the concrete. The reaction at the cathode is called a reduction reaction. A common reduction reaction is:
2H2O + O2 + 4e- → 4OH-
To maintain electrical neutrality, the ferrous ions migrate through the concrete pore water to these cathodic sites where they combine to form iron hydroxides, or rust:
2Fe2+ + 4OH- → 2Fe(OH)
This initial precipitated hydroxide tends to react further with oxygen to form higher oxides. The increases in volume as the reaction products react further with dissolved oxygen leads to internal stress within the concrete that may be sufficient to cause cracking and spalling of the concrete cover.
Corrosion of embedded metals in concrete can be greatly reduced by placing crack-free concrete with low permeability and sufficient concrete cover. Low-permeability concrete can be attained by decreasing the water to cementitious materials ratio of the concrete and the use of pozzolans and slag. Pozzolans and slag also increase the concrete resistivity thus reducing the corrosion rate even after it initiates. ACI 318-11, Building Code Requirements for Structural Concrete provides minimum concrete cover requirements that will help protect the embedded metals from corrosive materials. Additional measures to mitigate corrosion of steel reinforcement in concrete include the use of corrosion inhibiting admixtures, coating of reinforcement (for example, with an epoxy resin), and use of sealers and membranes on the concrete surface. Sealers and membranes, if used, have to be periodically reapplied.
Concrete and the Passive Layer
Although steel’s natural tendency is to undergo corrosion reactions, the alkaline environment of concrete (pH of 12 to 13) provides steel with corrosion protection. At the high pH, a thin oxide layer forms on the steel and prevents metal atoms from dissolving. This passive film does not actually stop corrosion; it reduces the corrosion rate to an insignificant level. For steel in concrete, the passive corrosion rate is typically 0.1 µm per year. Without the passive film, the steel would corrode at rates at least 1,000 times higher (ACI222 2001).

Because of concrete’s inherent protection, reinforcing steel does not corrode in the majority of concrete elements and structures. However, corrosion can occur when the passive layer is destroyed. The destruction of the passive layer occurs when the alkalinity of the concrete is reduced or when the chloride concentration in concrete is increased to a certain level.
The Role of Chloride Ions
Exposure of reinforced concrete to chloride ions is the primary cause of premature corrosion of steel reinforcement . The intrusion of chloride ions, present in deicing salts and seawater, into reinforced concrete can cause steel corrosion if oxygen and moisture are also available to sustain the reaction. Chlorides dissolved in water can permeate through sound concrete or reach the steel through cracks. Chloride-containing admixtures can also cause corrosion.
No other contaminant is documented as extensively in the literature as a cause of corrosion of metals in concrete than chloride ions. The mechanism by which chlorides promote corrosion is not entirely understood, but the most popular theory is that chloride ions penetrate the protective oxide film easier than do other ions, leaving the steel vulnerable to corrosion.

The risk of corrosion increases as the chloride content of concrete increases. When the chloride content at the surface of the steel exceeds a certain limit, called the threshold value, corrosion will occur if water and oxygen are also available. Federal Highway Administration (FHWA) studies found that a threshold limit of 0.20 percent total (acid-soluble) chloride by weight of cement could induce corrosion of reinforcing steel in bridge decks (Clear 1976). However, only water-soluble chlorides promote corrosion; some acid-soluble chlorides may be bound within aggregates and, therefore, unavailable to promote corrosion. Work at the FHWA (Clear 1973) found that the conversion factor from acid-soluble to water-soluble chlorides could range from 0.35 to 0.90, depending on the constituents and history of the concrete. Arbitrarily, 0.75 was chosen, resulting in a water-soluble chloride limit of 0.15 percent by weight of cement.
Although chlorides are directly responsible for the initiation of corrosion, they appear to play only an indirect role in the rate of corrosion after initiation. The primary rate-controlling factors are the availability of oxygen, the electrical resistivity and relative humidity of the concrete, and the pH and temperature.
Carbonation
Carbonation occurs when carbon dioxide from the air penetrates the concrete and reacts with hydroxides, such as calcium hydroxide, to form carbonates. In the reaction with calcium hydroxide, calcium carbonate is formed:
Ca(OH)2 + CO2 → CaCO3 + H2O
This reaction reduces the pH of the pore solution to as low as 8.5, at which level the passive film on the steel is not stable.
Carbonation is generally a slow process. In high-quality concrete, it has been estimated that carbonation will proceed at a rate up to 0.04 inches per year. The amount of carbonation is significantly increased in concrete with a high water-to-cement ratio, low cement content, short curing period, low strength, and highly permeable or porous paste.
Carbonation is highly dependent on the relative humidity of the concrete. The highest rates of carbonation occur when the relative humidity is maintained between 50 and 75 percent. Below 25 percent relative humidity, the degree of carbonation that takes place is considered insignificant. Above 75 percent relative humidity, moisture in the pores restricts CO2 penetration. Carbonation-induced corrosion often occurs on areas of building facades that are exposed to rainfall, shaded from sunlight, and have low concrete cover over the reinforcing steel.
Carbonation of concrete also lowers the amount of chloride ions needed to promote corrosion. In new concrete with a pH of 12 to 13, about 7,000 to 8,000 ppm of chlorides are required to start corrosion of embedded steel. If, however, the pH is lowered to a range of 10 to 11, the chloride threshold for corrosion is significantly lower—at or below 100 parts per million. Like chloride ions, however, carbonation destroys the passive film of the reinforcement, but does not influence the rate of corrosion.
Example of carbonation on a building facade. 
Dissimilar Metal Corrosion
When two different metals, such as aluminum and steel, are in contact within concrete, corrosion can occur because each metal has a unique electrochemical potential. A familiar type of dissimilar metal corrosion occurs in an ordinary flashlight battery. The zinc case and carbon rod are the two metals, and the moist paste acts as the electrolyte. When the carbon and zinc are connected by a wire, current flows. In reinforced concrete, dissimilar metal corrosion can occur in balconies where embedded aluminum railings are in contact with the reinforcing steel. Below is a list of metals in order of electrochemical activity:
1. Zinc 5. Nickel 9. Copper
2. Aluminum 6. Tin 10. Bronze
3. Steel 7. Lead 11. Stainless Steel
4. Iron 8. Brass 12. Gold
When the metals are in contact in an active electrolyte, the less active metal (lower number) in the series corrodes.
References
ACI Committee 222,
Protection of Metals in Concrete Against Corrosion, ACI 222R-01, American Concrete Institute, Farmington Hills, Michigan, 2001, 41 pages.
ACI Committee 318,
Building Code Requirements for Structural Concrete, ACI 318-05, American Concrete Institute, Farmington Hills, Michigan, 2005, 443 pages.
Clear, K.C., and Hay, R.E., “
Time-to-Corrosion of Reinforcing Steel in Concrete Slabe, V.1: Effect of Mix Design and Construction Parameters,” FHWA-RD-73-32, Federal Highway Administration, Washington, DC, April, 1973, 103 pages.
Clear K.C., “
Time-to-Corrosion of Reinforcing Steel in Concrete Slabs,” Federal Highway Administration, PB 258 446, Vol. 3, April, 1976.
PCA,
Types and Causes of Concrete Deterioration, Portland Cement Association, Skokie, Illinois, 2002, 16 pages.