
How Cement Is Made
Cement & Concrete
An English bricklayer made the first portland cement early in the 19th century by burning powdered limestone and clay on his kitchen stove. With this crude method, Joseph Aspdin of Leeds laid the foundation for an industry that annually processes literally mountains of limestone, clay, cement rock, and other materials into a powder so fine, it will pass through a sieve.
Portland cement is the basic ingredient of concrete. Concrete is formed when portland cement creates a paste with water that binds with sand and rock to harden.
Concrete is the most widely used resource in the world after water.
What Cement Is Made From
Cement is manufactured from materials heated together at high temperatures to form a rock-like substance that is ground into a fine powder.
Main Ingredients
- Calcium
- Silicon
- Aluminum
- Iron
Limestone, marl, and clay are commonly used sources of these primary elements.
Other Ingredients
Other materials used to manufacture cement include: shells, chalk, shale, slate, silica sand, iron ore, and blast furnace slag.
Events
The Cement Manufacturing Process
While each cement plant may differ in layout, equipment, and appearance, the general process of manufacturing portland cement is the same: crushed limestone and sand are mixed with ground clay, shale, iron ore, fly ash and alternative raw materials. Some manufacturers grind the raw materials dry, while others use a wet-grinding process involving water.
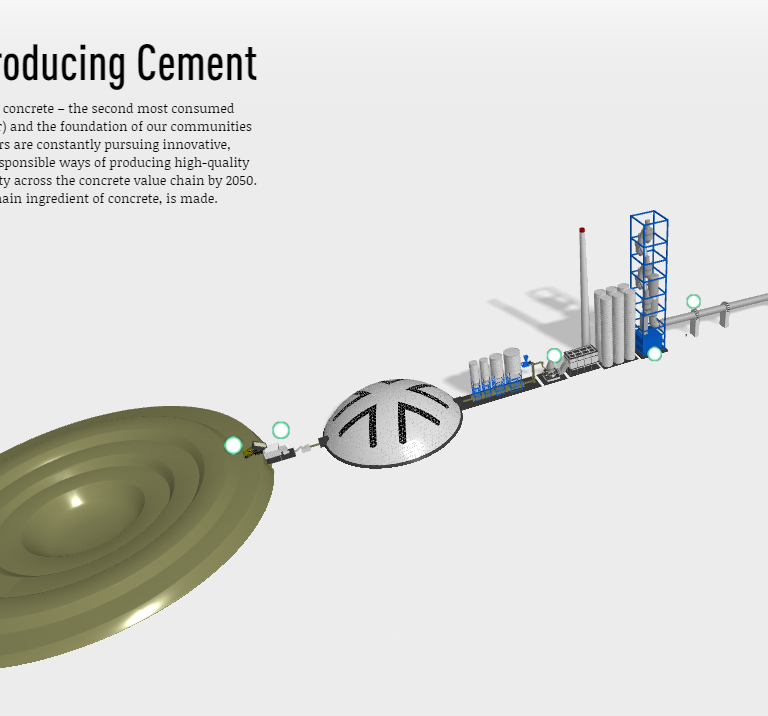
Sustainably Producing Cement
Cement producers are constantly pursuing innovative, efficient, and environmentally responsible ways of producing high-quality cement to realize carbon neutrality across the concrete value chain by 2050. Take a look at how cement, the main ingredient in concrete, is made.
Cement Goes Greener
The cement and concrete industry is dedicated to reaching carbon neutrality by 2050 or sooner. The use of lower-carbon cement blends, alternative fuels, re-use of waste products and other innovations is occurring and will continue to grow to lower the environmental impacts of concrete construction.



More to Learn
Got cement and concrete questions? We’ve got answers about the material, and how to use and care for it.










